By Jake Staples, Technical Team Leader, Ashland
The global flexible packaging market continues to evolve as shifts in consumer preference lean more toward convenience in single-serve or grab-n-go options, extended shelf life and sustainability. In addition to consumers driving innovation in flexible packaging, wide web and narrow web converters are beginning to converge into the packaging space as they see increased opportunity in equipment and coating technologies, thereby lowering the barrier to entry. With the advancements in ultraviolet light emitting diodes (UV LED) curing technology, opportunities in the flexible packaging space become more available to the converter base.
Flexographic packaging market trends
According to a Smithers Pira forecast, the global flexographic printing industry was expected to reach $980 billion last year, primarily driven by growth in packaging and labels.1 With more than 15,000 label converters and almost 500 flexible packaging converters globally – operating multiple press technologies in multiple plants – there is growing merger and acquisition activity that is converging the two market segments, convoluting the packaging space while strengthening a converter’s position. With the label market slated to grow only 2% annually and flexible packaging at a strong 5%, there is even more reason for a company to acquire adjacent technologies or competitors to complement its business strategy.
Many drivers are attributed to the growth in the flexographic packaging market, including consumer preferences (particularly from millennials, who desire convenience and single-serve packaging), the need for increased shelf life, pet food manufacturers moving away from multiwall bags to flexible packaging and growing interest in moving away from rigid to flexible structures.
Flexographic market segments
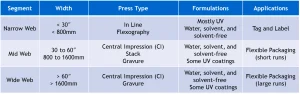
The flexographic market segments can be divided into three primary categories: narrow web, mid web and wide web, each defined in Table 1. Note: the widths defining each market segment are a general specification and not stated industry standard.
Due to the significant growth in flexographic flexible packaging, the limitations of traditional UV curing technologies that currently prohibit label and flexible packaging converters from participating in this segment have been exposed. This is particularly true in the food and beverage segment, which is more than 50% of the overall flexible packaging market. One way to eliminate the barrier is to fully understand the benefits of UV LED technology and the value it brings to the flexible packaging market.
Types of UV LED flexographic formulations and technologies
Inks, coatings and adhesives formulated for cure with UV LED are increasingly gaining traction in the narrow, mid and wide web markets. While some formulations – such as silicone-release and highly functional coatings – with smaller overall market demand have seen limited UV LED development, the broader portfolio of UV LED-formulated inks, coatings and adhesives already exist commercially for a wide range of converter requirements. In situations when off-the-shelf formulations are not the best match for a particular application, the existing formulation typically can be modified to fit the converter’s specific processing, construction or application needs.
The greatest commercial use today at press speeds of up to 1,000 fpm are UV LED formulated line, process and high-density inks, in both general purpose and low-migration formulations. Metallic and fluorescent UV LED variations also are being adopted by converters, since the use of UV LED curing sources results in a truer and brighter cured metallic and fluorescent look. In addition, fluorescents have been shown to fade less when passed under multiple UV LED lamps on a press than with a similar number of passes under conventional mercury lamps. Recent formulation improvements in clear UV LED primers, laminating adhesives and protective over-varnishes now are enabling nonyellowing in the final cure at increasing press speeds that are approaching those of UV LED cured inks.
Advantages of UV LED curing
While new narrow web presses sometimes are sold with hot air driers for water-based and solvent-based formulations, the majority are equipped with UV driers. This is due to the many operational and final product advantages that UV curing offers converters and brand owners.
For the mid and wide web flexo, gravure and coating market segments, presses are generally built to run water-based, solvent-based or solventless inks, coatings and adhesives. Converters often will run a UV-cured silicone release coating in nitrogen, a UV-cured protective over-varnish or a laminating adhesive in combination with these formulations. This is commonly done offline or at a specified distance from the solvent formulations in order to comply with appropriate explosion-proofing requirements. It is important to note that, due to the electrical design of UV LED sources and their compactness, UV LED curing offers the potential to convert many of these mid and wide web applications to UV and even run UV LED technology inline with solvent formulations.
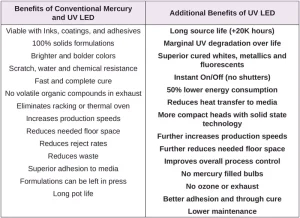
Regardless of the web width or press configuration, UV LED curing offers all the advantages associated with conventional mercury lamps, as well as additional environmental, operational and performance benefits (see Table 2).
UV LED laminating process
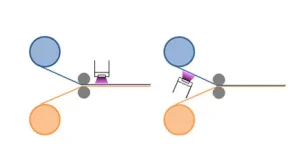
In flexible packaging applications, the process of laminating is used to securely bond two or more flexible constructions, such as PET, PE, PP, paper and foil, among others. The two constructions are unwound on press from their respective rolls. The laminating adhesive is applied to the substrate with lower absorption properties before being nipped or pressed to the second substrate. Formulation application methods include flexo, gravure and coaters. The two substrates, combined with the wet adhesive sandwiched between both layers, then are passed underneath the UV LED source for immediate cure (see Figure 1). Pressure-sensitive adhesives (PSAs), on the other hand, are dry cured, since the first construction and PSA are passed under the UV source before being pressed to the second construction.
Since UV LED has the advantage of transferring less heat to the pouch structures, it expands the range of substrate gauges that can be used in an application. It should be noted: This is not a cold cure technology, as UV LED wavelengths still are a form of energy. UV LED basically results in less energy being converted to heat at the substrate surface when compared to other curing sources.
For some incredibly heat-sensitive substrates, it is helpful to cure on the outside surface of a chilled drum or roller as a means of managing better substrate and process control, but it is not always necessary.
UV LED laminating adhesives vs. traditional UV laminating adhesives
In determining the potential value proposition of UV LED vs. traditional UV mercury technology, one should look to the higher performance of the laminating adhesives in conjunction with UV LED curing technology. With UV LED, the corresponding laminating adhesive provides a longer pot life and immediate cure, as compared to solvent-free laminating adhesives, which may take up to five days to fully cure. With the benefit of immediate cure, flexible packaging rolls can be slit and pouched immediately, offering the converter short runs and quick turns to their customers. In addition, UV LED laminating adhesives can be applied with existing flexographic or gravure cylinders, and UV LED stations can easily be retrofitted onto existing equipment, which equals lower total capital investment.
Low-migration regulatory compliance for flexible packaging
In complying with regulations in food safety, particularly when introducing UV LED laminating adhesives, utilization of the appropriate regulatory agency – Food and Drug Administration (FDA) or European Food Safety Authority (EFSA) – guidelines for migration and risk assessment is critical. For the US, relying on the FDA’s Toxicology and Chemistry guidelines is the most logical starting point. Following these guidelines in developing the needed migration protocol that simulates the foods and conditions of use for food packaging, in addition to determining the risk of exposure to chemical migrants, will protect converters from potential legal implications.
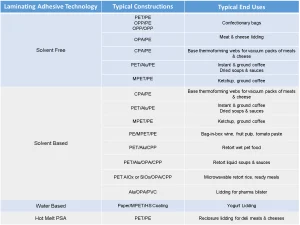
There are three areas that must be understood to evaluate safety in food packaging applications. First, it is necessary to understand the types of foods and ingredients that are going to be packaged. Table 3 shows the most common flexible packaging structures matched to typical end use applications and their corresponding adhesive laminating technology.
Secondly, once the application and the type of food that is being packaged have been identified using the FDA’s Toxicology and Chemistry Guidelines (see Table 4), it is necessary to determine the recommended simulants that will need to be used in condition testing to determine the migratory properties of the structure.

Last, consider the conditions of use to which the pouch will be subjected during the processing phase. Using the FDA conditions, testing must be completed per the appropriate condition for the required application. According to the FDA, the various conditions to be potentially tested are as follows:
- High temperature, heat sterilized or retorted [ca. 121ºC (250ºF)]
- Boiling water sterilized (100ºC)
- Hot filled or pasteurized above 66ºC (150ºF)
- Hot filled or pasteurized below 66ºC (150ºF)
- Room temperature filled and stored (no thermal treatment in the container)
- Refrigerated storage (no thermal treatment in the container)
- Frozen storage (no thermal treatment in the container)
- Frozen or refrigerated storage: ready prepared foods intended to be reheated in container at time of use
- Irradiation (ionizing radiation)
- Cooking at temperatures exceeding 121ºC (250ºF)
Another key aspect to consider when evaluating the structure is the type of functional barrier being used. The functional barrier prevents a varying degree of migration of the nonfood contact material in what is being pouched – particularly if the contents are food. However, there is little guidance from the FDA or European Union that defines performance of an adequate functional barrier and its corresponding acceptable migration levels.2 Ultimately, it is the converter and contract packager’s responsibility to determine the needed functional barrier. When testing the structural compliance of the package, ensure the hazard information of the migrants and safe dietary levels are reviewed. The FDA default exposure values can be used to determine the exposure level of the package, such as percent of diet exposed to the packaging type. This can be calculated by dividing the safe dietary level by the exposure where a detection limit can be derived.
The same FDA regulatory requirements that apply to today’s packaging standards also are applicable when transitioning from a traditional laminating adhesive, such as solvent-free, to UV LED. As long as the appropriate steps are taken in the risk assessment of the packaging – including component migration, toxicological data established limits and performing tests in all applicable food types, temperatures and conditions – UV LED technology is a viable, efficient and green method of cure.
Furthermore, UV LED technology has the advantage of providing a curing process that is reliable, repeatable and extremely controllable. With low migration and regulatory compliance, it is critical that process variables are monitored and kept within the defined operating window.
UV LED technology, coupled with UV LED-formulated laminating adhesives for low-migration applications, represent an innovative and sustainable technological trend that will have growing importance in flexible packaging for those early adopters searching for market differentiation in the narrow, mid and wide web space.
Experimental
Laminations were made on the Mark Andy P5 press with a Mark Andy Gen 2 ProLED (Phoseon FP601), 20 W/cm², 395 nm, flat glass emitting window, UV LED curing station. The flexographic anilox for applying the adhesive was 360 lpi/5.42 bcm. Multi-Plastics (MP) 2 mil PE was the base, and MP 75 gauge PP was the overlaminate. The laminating adhesives tested were Ashland Adhesive 1 and Adhesive 2. The press was run up to 500 fpm and at LED power settings ranging from 25% to 100%. Neither adhesive displayed any yellowing. The best adhesive bonds for this construction were achieved by Adhesive 1, so these laminations were chosen for migration testing. Initial hand peels resulted in destructive bonds up to 500 fpm at 25% power.

The migration testing was conducted by placing the laminations in specially designed aluminum and glass cells (see Figure 2), with the food contact side of the construction exposed to the food simulant. The food simulant was 95% ethanol to cover fatty food types, and 10 ml/sq.in. was applied as the default amount. Condition of Use C (hot fill above 150°F) was tested by holding the food simulant in the cells at 66°C for 2 hours, followed by 10 days at 40°C. HPLC-MS analysis was performed to detect any components of the adhesive in the food simulant, and the data were reviewed by a food packaging toxicologist, who performed a risk assessment. A safe threshold of 50 ppb was set by taking into account toxicological data and daily consumption factor of 1% used in Food Contact Notification (FCN) 642.
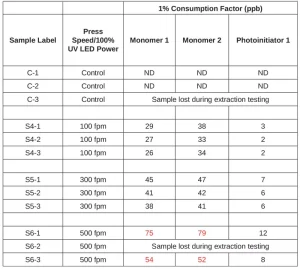
The data are presented in Table 5. These results indicated that the samples made at up to 300 fpm are safe for use in food packaging. The laminations made at 500 fpm had migration of both monomers above the limit.
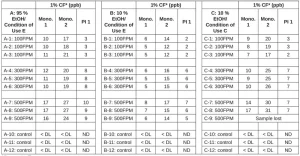
A second round of laminations were made in the lab. A Harper flexographic hand proofer with a 260l pi/5.48 bcm anilox was used for applying the adhesive to the samples, and they were cured with an American UV fitted with a Heraeus 16W/cm², 395 nm, UV LED at 100% power. MP 3 mil PE was the base, and MP 75 gauge PP was the overlaminate. The laminating adhesive tested was Adhesive 1. The conveyor belt was run up to 500 fpm. Migration testing was conducted by placing the laminations in aluminum and glass cells with the food contact side of the construction exposed to the food simulant. The food simulants were 95% ethanol to cover fatty food types and 10% ethanol to cover aqueous food types and 10ml/sq.in. was applied as the default amount.
Three conditions were tested. Condition of Use C (hot fill above 150°F) was tested by holding the food simulant in the cells at 66°C for two hours, followed by 10 days at 40°C. Condition of Use E (room temperature fill and storage with no thermal treatment) was tested by holding the food simulant in the cells at 40°C for 10 days. HPLC-MS analysis was performed to detect any components of the adhesive in the food simulant, and the data were reviewed by a food packaging toxicologist who performed a risk assessment. A safe threshold of 50 ppb was set by taking into account toxicological data and daily consumption factor of 1% used in FCN 642. The data are presented in Table 6. These results indicated that all the samples are safe for use in food packaging.
Conclusion
Under the right conditions of adhesive chemistry, degree of cure, laminate construction, food type and condition of use, UV LED laminating adhesives are safe for use in flexible food packaging. It is important for converters to confirm the safety of the adhesive used in the flexible packaging. This can be achieved by careful adhesive selection and control of manufacturing practices. Suppliers of UV LED curing stations and laminating adhesives can assist in this process.
Acknowledgements
The author acknowledges Dr. Joseph Spinnato of Ashland for his work on the migration risk assessment, Jennifer Heathcote of Eminence UV for her advice during the press trial and Catherine Heckman of Ashland for her assistance. The trial was supported by FlintGroup through the use of its pilot press.
References
- Smithers Pira, 1/2014, Global printing market to top $980 billion by 2018, <www.smitherspira.com/news/2014/january/global-printing-market-to-top-980-billion-by-2018>
- FDA, 12/14/2017, Packaging & Food Contact Substances (FCS), <www.fda.gov/Food/IngredientsPackagingLabeling/PackagingFCS>
Jake Staples, technical team leader for Ashland, specializes in radiation curable coatings, primers and adhesives, with more than 17 years of energy-curable expertise – particularly within the development of UV LED and EB technologies. His roles include product development, application support and technical sales. He has presented at industry conferences, including RadTech events. He earned a BA in physics from Carthage College and an MS in mechanical engineering from the University of Wisconsin-Milwaukee.






