In the last edition of “Professor’s Corner,”1 a novel method for reducing the viscosity of a common acrylate-functional epoxy-based oligomer was introduced. This method was developed over several years by undergraduate students at the University of Houston-Downtown and involved the use of deionized water (DI H2O) as a diluent. The DI H2O was found to be more effective at reducing the viscosity of the oligomer than were two of the most effective acrylate-functional monomers, octyl/decyl acrylate (ODA) and 1,6-hexanediol diacrylate (HDODA)2, 3. At only 2% by mass, the DI H2O produced a significantly lower viscosity than did either of the two monomers at 4%! (See Figure 4 in Reference 1.)
Investigating Compatibility of DI H2O-Containing Oligomer with Monomers
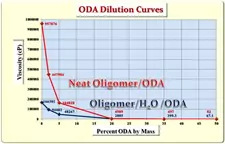
Having demonstrated that DI H2O was a very effective diluent for this oligomer, a second phase of the investigation was initiated to determine whether the presence of quite polar DI H2O at 2% concentration would create formulating problems with relatively nonpolar monomers. HDODA and ODA were, once again, chosen for this part of the study because of their relatively nonpolar molecular structures. HDODA is a difunctional monomer containing a 6-membered nonpolar hydrocarbon chain, while ODA is an equimolar mixture of two monofunctional monomers consisting of an 8-membered and a 10-membered hydrocarbon chain, respectively. The study involved formulating the neat oligomer and separately, the water-containing oligomer, with varying concentrations of ODA and HDODA up to 50% by mass. Figure 1 depicts the viscosity results of this study for ODA while Figure 2 demonstrates the results for HDODA.
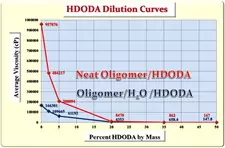
The first thing to note is that, with both monomers, the formulations were completely free of haziness or other indications of monomer/water incompatibility to levels as high as 50% monomer. The relatively nonpolar nature of these two monomers might lead one to expect a compatibility problem with the water-containing oligomer, but none was observed. Also, a close inspection of the data in Figures 1 and 2 reveals that, for all monomer concentrations up to 50%, the water-containing oligomer/monomer mixtures were lower in viscosity than were the corresponding neat oligomer/monomer formulations. Thus, for formulators, the DI H2O-containing oligomer not only provides improved ease of handling, but also potentially minimizes the concentration of (meth)acrylate monomers necessary to produce the desired end-use properties.
Investigating Properties of Formulated Systems
In a third part of the study, student researchers prepared comparative formulations containing 70% by mass oligomer and 30% monomer. The neat epoxy-based oligomer was used in half of the formulations, and the oligomer containing 2% DI H2O was used in the other half. ODA was used at 30% in one set of formulations, 30% HDODA was used in a second set, and a 15/15 blend of both monomers was used in a third set. Each formulation prepared also contained one of two separate photoinitiators (PIs) at 3.0 pph. Thus, the study involved a total of 12 formulations. The 12 formulations were prepared, viscosities were measured, and polymer films were UV-polymerized and characterized using dynamic mechanical analysis (DMA). Experimental details for this study are given in Reference 3. A summary of the results follows.
25° C Viscosity. The trends in 25° C viscosities of the formulations were just as expected. All six DI H2O-containing formulations showed significantly lower viscosities than their corresponding neat oligomer-containing formulations, regardless of the monomers or PI used. Also, as the ODA concentration was systematically decreased and replaced by HDODA, the viscosities increased. Finally, the viscosities of formulations containing the PI, 2-hydroxy-2-methyl-1-phenyl-propane-1-one, were consistently lower than those containing 1-hydroxycyclohexyl phenyl ketone PI. The former PI is a liquid while the latter PI is a solid.
Glass Transition Temperatures (Tgs). The Tgs of the films were determined from the peak of the tan δ curve in the DMA scans. As the monofunctional ODA was replaced by the difunctional HDODA, the average functionality of the formulations increased. Thus, it was expected that the Tgs of the formulations also would increase with increasing functionality. This trend was observed for all formulations, regardless of the oligomer or PI being utilized.
A bit more surprising were the Tg results when comparing the formulations containing the neat oligomer with those of the DI H2O-containing oligomer and the same monomer package or PI. At 30% by mass ODA, the Tgs of all four formulations essentially were the same (within 3° C). At 15/15 ODA/HDODA and 30% HDODA, however, some differentiation was observed. While some of the differences were small, and most likely not statistically significant, the larger differences did indicate higher Tgs for the water-containing formulations. This is unexpected since the formulations containing DI H2O were slightly lower in overall average functionality, leading one to assume (perhaps falsely – see below) that the cross-link density would be slightly lower also. Further, the DI H2O could act as a plasticizer. Perhaps the lower viscosity observed for the water-containing formulations allowed for more component diffusion during polymerization, causing an increase in reactivity and producing a higher conversion of monomers and the oligomer to polymer. Also, since DMA is a thermal analytical method, water may have been driven out of the films during testing, reducing its apparent plasticizing effect.
Other Thermomechanical (DMA) Properties. In addition to Tg data, the researchers also used DMA to determine the relative cross-link densities of the films as indicated by the storage modulus readings in the “rubbery plateau.” This is the region in a DMA scan above the Tg where the storage modulus becomes independent of temperature4. It is understood that the higher the value of the storage modulus in this region, the higher the cross-link density of the film is expected to be. Not surprisingly then, when the average functionality was increased by increasing HDODA in the formulation, the apparent cross-link density also increased with both oligomers. However, in contrast to some of the Tg data that indicated the water-containing systems to have marginally higher Tgs, the apparent cross-link densities for some of these formulations were significantly lower than for the neat oligomer-based formulations. This was true for the formulations containing either 15% or 30% HDODA, along with the liquid PI. The storage moduli in the rubbery plateau of the water-containing formulations were lower than those containing the neat oligomer by about 16 and 19 MPa, respectively. For the formulation containing 30% ODA, the opposite was true, but only slightly. Since ODA is monofunctional, it cannot contribute to cross-link density, so the difference observed (< 3 MPa) was probably not statistically significant. For those containing the solid PI, the results tended to be opposite, indicating a possible PI effect. However, the relatively low cross-link density of these formulations overall may mean that the observed differences are not significant. Clearly, for a better understanding of these effects, further research is needed.
Conclusions
In this and the last edition of “Professor’s Corner,” a previously reported simple method for reducing the viscosity of an acrylate-functional epoxy-based oligomer was discussed2, 3. This method involved adding 2 g of DI H2O to 98 g of the oligomer. The water reduced the viscosity of the oligomer by nearly 89%! The investigators also found that the water-containing oligomer was quite compatible with two of the more nonpolar acrylate-functional monomers and that the thermomechanical properties (DMA) of the UV-polymerized films were comparable to those of identical formulations based on the neat oligomer. While substantially improving the handling characteristics for both oligomer suppliers and formulators, this inexpensive process also has the potential to reduce the amount of acrylate-functional monomers needed to achieve desired properties for formulated products. This approach to oligomer viscosity reduction seems to offer real benefits to oligomer suppliers and formulators alike. More research in this area is needed and encouraged.
 Byron K. Christmas, Ph.D.
Byron K. Christmas, Ph.D.
Professor of Chemistry, Emeritus
University of Houston-Downtown
b4christmas@gmail.com
References
- Christmas, B., “The Problem of High Viscosity Oligomers – Part 1”, UV+EB Technology, Vol. 7, No. 2, Quarter 2, 2021, pp. 16-17.
- Christmas, B., “Reducing the Viscosity of an Epoxy-based Oligomer Using Minimal Amounts of Aqueous Electrolyte Solutions: Revisiting the Use of Water as a Diluent for UV-Polymerizable Oligomers”, Photopolymerization Fundamentals Conference 2011, Breckenridge, CO, June 27, 2011.
- Baig, N., Truong, M., Dumas, T., and Christmas, B., “A Simple Process for Preparing Low Viscosity Diacrylate-Functional Epoxy Oligomers”, Proceedings, RadTech UV & EB Technology Expo & Conference, Chicago, IL, April 30-May 2, 2012.
- Christmas, B., “Understanding Glass Transition Temperature: Part 2” UV+EB Technology, Vol. 6, No. 2, Quarter 2, 2020, pg. 13.
- Morales, T., Rueda, E., and Christmas, B., “The Relative Effects of AC- and DC-Powered UV Lamp Systems on Properties of UV-Polymerized Films”, RadTech 2008 UV&EB Technical Conference Proceedings, RadTech International North America, May 2008.