By Chris Orilall, Zack Weinert, Jon Scholte, Chuck Dong, and Jeff Klang, Arkema Inc., Sartomer Business Unit
Abstract
Energy-curable pressure sensitive adhesives (EC-PSAs) eliminate the need for drying, solvent extraction or preheating steps (compared to traditional waterborne, solventborne and hot melt methods). These processing benefits make such adhesives particularly suitable for temperature-sensitive substrates and in-line application. Here, we demonstrate structure-property relationships between PSA formulation and properties such as tack, peel strength, shear resistance and temperature performance. Highlights include screening the viscoelastic properties and investigating routes to higher shear adhesion failure temperature (SAFT) PSAs.
Introduction
Adhesives are nonmetallic materials used to bond other materials by adhesion to their surfaces and cohesion within the adhesive layer. Two different processes describe most adhesive bonding: 1) the adhesion phenomenon or 2) by PSA action. In the former, adhesive fluid is transformed into a solid upon bonding. A PSA, however, conserves its fluid state after its bond building. Therefore, some notable benefits of PSAs are that the resistance to de-bonding is moderate and the joint may be delaminated without destroying the laminate components.1
Today, the three main PSA technologies are waterborne (WB-PSA), solventborne (SBPSA) and hot melts (HM-PSA). The global demand for these types of PSAs totals ~ 93%, with the rest being UV-curable hot melts (UVHM-PSA) and silicone-based PSAs.2 Broadly speaking, SBPSAs and UVHM-PSAs tend to offer “high” performance, HM-PSAs offer medium performance and WB-PSAs offer lower performance at the lowest cost. Each of these technologies has its advantages, but also distinct disadvantages: low solvent/plasticizer resistance for HM-PSAs, high concentration of volatile organic compounds (VOC) for SB-PSAs and large equipment footprints required for WB-PSAs.
Herein, we present a UV-curable or electron beam (EB)-curable PSA made from 100% solids, i.e., no solvents. These energy-curable systems are sometimes referred to as EC-PSA “syrup” formulations. The product can be applied as a viscous liquid system without the need for heat (depending on the viscosity requirements of the coating unit). Without solvents or water, this product eliminates the need for highly regulated solvent extraction systems or for time-consuming oven-drying steps. As a result, EC-PSAs can lead to higher productivity and lower manufacturing costs vs. conventional PSA products. Additionally, rapid cure, improved dimension accuracy, heat resistance, chemical resistance and heavy coating weights are possible. To use EC-PSAs, existing lines can be retrofitted with a UV lamp, a UV-LED lamp or an EB unit. Additionally, EC-PSA syrups provide ideal formulation platforms for cure-in-place (CIP) automated applications. As will be discussed, the viscosity of the EC-PSA formulation can be adjusted by varying the ratio of components, thus tailoring the formulation for use in any deposition system. More so, this approach allows for control over the performance of the PSA through choice and composition of starting ingredients.
The main components in a typical EC-PSA formulation areas follow:
- Oligomers: Provide much of the shear strength and also affect tack, peel, reactivity, creep resistance, heat resistance and chemical resistance.
- Monomers: Mainly serve as reactive diluents, i.e., lower the viscosity of the formulation. These components also control surface wetting, leveling and other physical properties.
- Photoinitiators: Absorb UV energy to produce free radicals that induce polymerization. Formulations intended for EB curing do not need photoinitiators to polymerize.
- Additives
- Stabilizers prevent premature curing due to low-level light exposure in storage.
- Tackifiers improve the pressure-sensitive nature or “stickiness” of the adhesive.
- Other additives include adhesion promoters, fillers, antioxidants, plasticizers, color pigments, dyes, defoamers, flatting agents, wetting agents and slip aids.
In general, the most fundamental material properties governing EC-PSA performance are3:
- Rheology
- Study of the change in form and flow of a material. It generally is applied to viscoelastic materials.
- Uncured state: Rheology is important with regard to application and coating.
- Cured state: PSA material must have sufficient flow to provide wetting and tack, yet have enough resistance to stress to provide for high adhesive strength.
- Molecular Weight
- PSAs are based on very high-molecular-weight rubber polymers, which restrict flow by molecular entanglement.
- When high strength, heat resistance and chemical resistance are required, the molecules must be chemically crosslinked to provide for a three-dimensional network structure.
- Acrylate Functionality
- Number of unsaturated sites per molecule.
- Crosslink density increases with the functionality of the resin, all else equal.
Additionally, the type of substrate is critical to EC-PSA performance. In general, metals have high surface energy and tend to result in high peel strengths, while plastics have low surface energy and tend to result in lower peel strengths. For plastics, treated surfaces tend to improve PSA performance (versus untreated surfaces).
Experimental details
Sample preparation. PSAs were typically coated onto a 50 µm untreated PET facestock using an automatic film coater (MTI Corp. MSK-AFA-II). The thickness of the liquid PSA coating was controlled by a combination of the viscosity of the formulation, the gap of the coater (adjusted by a micrometer screw gauge) and the speed of the automatic coater. The liquid PSA coating was then cured by using one or multiple passes under one or two 400 W/in mercury vapor lamps (Inpro system) with a speed of 50 ft/min. The effective energy density (mJ/cm2) was measured using a power puck (EIT UV Powerpuck II). The PSAs also were cured using UV-LED lamps (365 nm or 395 nm). After curing, the samples were laminated onto silicone release paper, and 1-inch strips were cut for testing. For EB curing, the liquid PSA coatings were cured using an ESI eBeam unit at 50 ft/min, typically between 5 to 15 Mrad and 175 kV with just one pass.
180° peel strength. The PSA peel samples were prepared by applying 1-inch-wide PSA strips to stainless steel or polypropylene panels (ChemInstruments) using two passes with a 4.5-pound automatic roller. The PSA-laminated samples and panels were stored in a constant humidity room (at 72°F and 60% humidity) for a predetermined dwell time (one hour, one day or three days) before testing. The peel strengths were measured at a 180° angle with a peel speed of 12 in/min following the ASTM-D903-98 standard using a tensile tester (Instron 5543).
Probe tack. The probe tack was measured following ASTM-D2979-95 using a probe tack tester (ChemInstruments PT-500).
Shear resistance. Shear resistance was tested following ASTM 4498-95. One square inch of PSA was applied to a stainless steel panel using two passes with a 4.5-lb automatic roller. The PSA and panel were allowed a dwell time of one hour before testing. A mass of 1 kg or 2 kg was then attached to the other end of the PSA strip. A shear tester (ChemInstruments Shear-10) was used to hold the samples vertically while the mass suspended and to record the time required for the PSA strip to fully detach from the substrate. The shear resistance is reported as this time in minutes.
Shear adhesion failure temperature (SAFT). SAFT test samples were prepared in a similar manner to shear resistance samples. One square inch of PSA was applied to a stainless steel panel using two passes with a 4.5-lb automatic roller. The PSA and panel were allowed a dwell time of 1 hour before testing. A 1 kg mass was then attached to the other end of the PSA strip. A shear tester housed in an oven was used to perform the test. The starting temperature (T0) was noted (usually room temperature) and the oven temperature ramp (ramp rate of 1°C/min) and timer were started simultaneously. When the mass fell, the SAFT value for that PSA sample was calculated as: ramp rate (°C/min) x time (min) + T0 (°C).
Rheology. Dynamic mechanical analysis experiments were performed using a rheometer (TA Instruments RDA111) at a frequency of 1 Hz and heating rate of 3°C/min. All samples were tested using 8 mm parallel plate applying a static compressive force and tested in shear mode in the linear viscoelastic region. The peak tan δ temperature and storage modulus G’ at 20°C are reported. Frequency sweeps were done at 30°C in RDA 111, using 8 mm parallel plates from 0.01 rad/s to 100 rad/s.
Results and discussion
In this paper, we investigated the role of oligomers, monomers, photoinitiators and curing on EC-PSA performance.
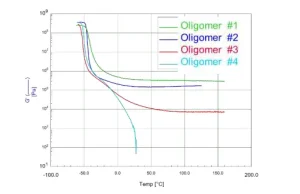
Choice of oligomers. Previously, we described how rheology can be used to down-select oligomers for use in PSAs.4 Here, the Dahlquist Criterion specifies that for measurable quick tack, the elastic modulus must be below a certain fixed value that is fairly independent of the nature of the adhesive, the adherend and the applied pressure. In the case of PSAs, tack ceased when the storage modulus (G’) was greater than 105 Pa.5,6 A typical DMA screening of four commercially available oligomers is shown in Figure 1. Here, Oligomers 1 and 2 would not be suitable for use in PSAs because G’ is greater than 105 Pa. Oligomers 3 and 4 both meet the Dahlquist Criterion for PSAs. Oligomer 3, however, would be most suitable because of its ability to flow while maintaining some structural integrity.
Choice of monomers. The coating unit used in the PSA application often sets the viscosity limits of the liquid formulation. This constraint, in turn, dictates the amount of monomer(s) that go into the final EC-PSA formulation. Many monomers have viscosities as low as 10 cP. The choice of acrylate monomer depends on the final PSA performance being sought, i.e., type of substrate, peel strength, shear, tack, application temperature of liquid PSA, service temperature of cured PSA, removable/permanent use, etc. In general, increasing the monomer functionality leads to higher cure speed, higher Tg, higher crosslink density, higher shear strength, greater chemical resistance, greater thermal resistance and lower flexibility. Often a combination of monomers of varying Tg is used to tailor properties.
Certain trends were observed for the EC-PSA in its cured state. When the adhesive Tg was greater than the service temperature, the PSAs were rigid and, to some extent, brittle. They had high shear and temperature resistance, but low impact and peel strengths; high barrier properties against chemicals and water – along with low water uptake and swelling – but poor resistance to thermal cycling. They were prone to crack propagation and had low thermal expansion coefficients. Conversely, the opposite effects were observed when the Tg of the EC-PSA was less than service temperature.
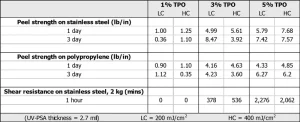
Choice of photoinitiator. To investigate the effects of photoinitiator concentration in an EC-PSA formulation, we varied the diphenyl(2,4,6-trimethylbenzoyl)phosphine oxide (TPO) loading from 1% to 5% into a base UV PSA formulation (CN 9073). The liquid formulation was cured at 200 mJ/cm2 (low cure, LC) and 400 mJ/cm2 (high cure, HC). The thickness of the cured PSA was held constant at 68 µm. Table 1 shows the results of the PSA performance evaluation that followed. The testing included peel strength on stainless steel and polypropylene at different dwell times, and shear resistance using a 2 kg mass. At 1% TPO concentration, we observed some adhesive performance but no shear resistance – most probably an indication that the formulation was undercured. We noticed that the shear resistance increased from ~500 mins to ~ 2000 mins for formulations made with 3% TPO and 5% TPO respectively. Not surprisingly, we expect that this is due to greater degree of cure for 3% TPO vs. 5% TPO. The effect of photoinitiator concentration on peel strength is less evident as other factors – such as dwell time, energy dosage and thickness – play larger roles. However, overall better performance was seen with 5% TPO vs. 3% TPO.
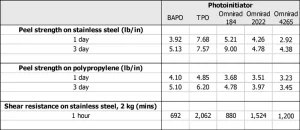
We also investigated the choice of photoinitiator on PSA performance. Here, we investigated PSA performance of a base UV PSA formulation (CN 9073) with 5% loading of either 2,4,6-trimethylbenzoyl-diphenyl phosphine oxide (TPO), Omnirad® 184, Omnirad® 2022 or Omnirad® 4265 and with 3% loading of phenylbis 2,4,6-trimethylbenzoyl-phosphine oxide (BAPO) (to standardize amount of radicals generated). PSA performance for formulations with these different photoinitiators is captured in Table 2. (PSA adhesives were all 68 µm thick and cured using H bulb at 400 mJ/cm2). The results indicate that the choice of photoinitiator is crucial to PSA performance and is dependent on the other components in the formulation and on curing conditions.
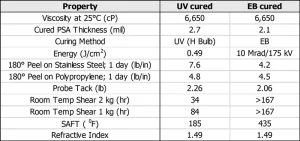
UV PSA vs. EB PSA. We investigated the PSA performance of a base energy-curable formulation (CN 9073) cured via UV and EB (Table 3). For the UV-curable formulation, 5% TPO was employed, while no photoinitiator was required for EB curing. Just as different adhesive properties can be accessed within UV curing alone, different PSA performances were observed between the two curing methods. The main advantage of EB curing was higher shear and unprecedented high SAFT. We also have seen high PSA performance when curing using UV-LED lamps (365 nm and 395 nm) comparable to that shown in Table 3, but this finding has not been fully investigated.
Versatility of EC-PSAs
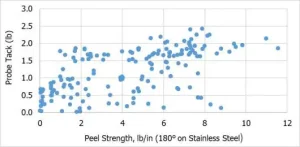
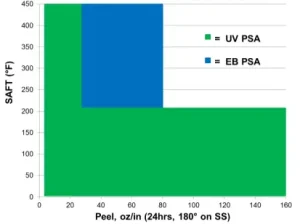
Figure 2 captures the range of PSA performance (tack and peel) achievable to-date via UV curing. While this figure does not capture the viscosity of each data point, it does demonstrate how EC-PSAs can be tailored to meet very specific PSA application targets. One interesting advantage that EC-PSAs may be able to offer is low tack with high peel strength, which could potentially be useful for applications where minimal pressure is required for bonding. Furthermore, Figure 3 shows the range of SAFT values that can be achieved by either UV PSA or EB PSA. In the case of EB-cured PSAs, SAFT values of > 400°F are unprecedented, and current research and development efforts are focused on extending the achievable property spaces of both UV- and EB-PSAs, e.g., UV-PSAs with high SAFT and high peel strength. We have attributed higher SAFT values for EB PSAS to the higher acrylate conversion for EB cure vs. UV cure, which directly results in higher crosslinking density for EB PSAs vs UV PSAs.
Conclusion
In summary, we found that a number of variables affect EC-PSA properties, including rheology and chemical characteristics. The relationships between different measures of adhesive performance were analyzed, as were different methods of curing PSAs. Specifically, the choice and amount of oligomer, monomer, photoinitiator and additives are all crucial to the starting point liquid formulation. With this wealth of available chemistry, the range of achievable EC-PSA performance is quite broad, and the ability to tailor each formulation to specific performance targets makes this approach very versatile. υ
References
- Benedek. Pressure-Sensitive Adhesives and Applications, 2nd ed. (2004) New York, NY. Marcel Dekker, Inc.
- AWA Global Specialty Pressure-Sensitive Tape Market Study 2016.
- Petrie, Edward M., “Formulating UV-Curing Liquid Pressure-Sensitive Adhesives,” Adhesives and Sealants Industry Magazine. November 2008.
- J. Lu and C. Dong. Radiation Curable Pressure Sensitive Adhesives. Radtech 2016.
- “Viscoelastic Properties of Pressure Sensitive Adhesives,” The Journal of Adhesion, 60:1-4, 233-248.
- C. A. Dahlquist. Proc. Nottingham Conference on Adhesion. Part III, Chapter 5, p. 134.
Acknowledgments
The authors want to thank Zachary Hoffman, Kejun Chen and the rest of the research and development team at Sartomer. We also thank Sara Reynaud of the Arkema Analytical department for training, support and advice concerning the rheology of PSAs. We thank our PSA counterparts at Bostik and Arkema Coating Resins. We would also like to thank Im Rangwalla at Energy Science Inc. (ESI) for the coating and curing of EB-PSA samples.





