Adhesion, or lack of it, is one of the most critical attributes for most coatings, inks and, of course, adhesives themselves. While the concept of adhesion may sound simple, the details can be more complex.
A key factor for most types of adhesive bonding is substrate wetting, which refers to whether the coating flows over the substrate or whether it “beads up.” If the coating or ink does not fully cover the surface of the substrate, then the adhesion will be limited by the diminished contact area. There are several ways a formulator can affect the wetting out of the formulation on the surface of the substrate. These methods can be summarized in the relationship between substrate surface energy and coating surface tension.
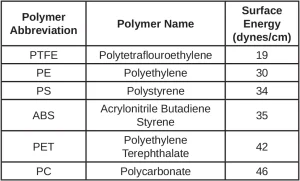
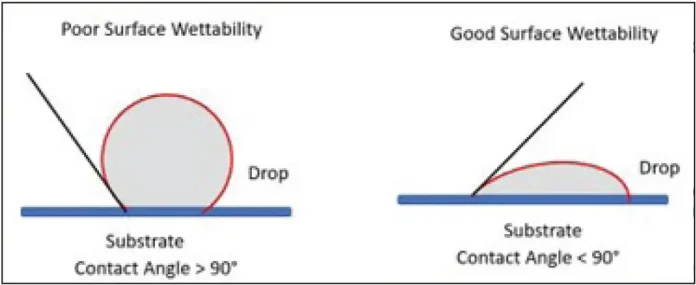
To ensure good wetting or coverage of the formulation on the material surface, the coating surface tension must be less than the substrate surface energy – generally, the surface energy of the substrate should be five to 10 dynes/cm greater than coating. This is illustrated in Figure 1, which shows a diagram of the contact angle of a drop on a surface. For adequate wetting, one wants to have a minimum contact angle, and an obvious way to accomplish this is to select a substrate with a high surface energy. Table 1 illustrates the surface energies for some common plastics. Note: Metals tend to have higher surface energy than plastics, which frequently make them easier to wet out by the coating
or ink.
From a practical point of view, the formulator does not always have the option to select a substrate with a sufficiently high surface energy as the substrate usually is determined by the application. However, surface energy of a plastic frequently can be increased by surface treatment on the coating line by either a corona, flame or plasma treatment.
Another option is to decrease the coating surface tension of the formulation, most frequently done through the addition of additives. A wide range of commercially available additives for energy-curable formulations can modify the surface tension of the formulation. As these additives frequently are used at small levels, there is only a small probability that the additives will influence the cured formulation properties.
Regardless of whether the best option is to increase the substrate surface energy or decrease the coating surface tension, it is important that the surface of the substrate is clean of oils and greases that can interfere with the bonding of the coating or ink. For plastics, mold release agents on the surface also can cause problems. If contaminants do adversely affect adhesion, this could be remedied by a simple wipe-down of the problem surface with a suitable organic solvent, such as isopropyl alcohol.
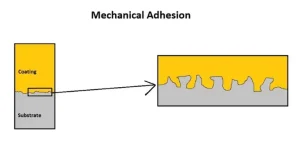
When it comes to modifying the adhesion properties of a coating or ink, there are usually only two common mechanisms of adhesion considered when formulating energy-curable coatings: mechanical bonding and chemical bonding. Mechanical adhesion occurs when adhesive materials physically lock into the crevices of the surface. This requires that the substrate surface be physically abraded or chemically etched, unless the substrate has a naturally rough surface (Figure 2).
Physically abrading or chemically etching a surface tends to work best with metal substrates as certain plastics do not respond well to this type of surface preparation. For plastics, a method that can work well is to treat the surface with an appropriate organic solvent. This will swell the surface of the substrate, allowing the coating or ink to grab onto the now-roughened surface.
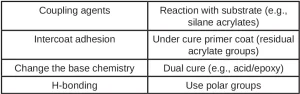
Another type of adhesion formulators can use to their advantage is that of chemical adhesion or bonding. Chemical adhesion occurs when the coating chemically bonds with groups on the surface of the substrate. By bonding to the substrate, the coating or ink essentially becomes part of the substrate, thus minimizing any tendency for the coating or ink to be easily removed. Table 2 shows a selection of typical routes for using chemical bonding.
Another way to modify adhesion through a chemical route does not involve chemical bonding to the substrate, and that is to significantly change the UV-curable chemistry of the formulation itself. The shrinkage for (meth)acylates upon curing can be as much as 20%. A cause of failure of adhesion for many free radical type formulations is the physical pulling away of the formulation as it cures due to shrinkage of the reactive components. If the base chemistry of the formulation is free radical cure, it is possible to greatly improve the adhesive properties of the formulation by switching to cationic cure. As a result of the mechanism of cationic cure, shrinkage is dramatically reduced. This, unfortunately, is not an easy fix for improving the adhesion of a formulation as there may be new problems created if the properties, cost, cure speed, etc. of the new cationic cure formulations do not meet the requirements of the coating or ink. In selected cases, a hybrid system composed of a mix of free radical and cationic cure may result in sufficiently increased adhesion, along with acceptable properties to meet the needs of the target application.
Chemical adhesion can be improved using coupling agents, such as silane acrylates. This class of materials acts as a bridge between the substrate surface and the coating or ink. They typically will have silane functionality that will react with hydroxyl groups on the substrate surface. The other end of the molecule usually will contain an acrylate or methacrylate group, and this UV reactive end group will take part in the photopolymerization and bind the UV-curable photopolymer to the surface.
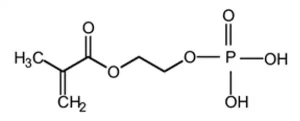
In addition to the use of silanes, other types of adhesion promoters are commonly used in UV-curable formulations. The most widely used adhesion promoter for metal substrates is 2-Hydroxyethyl methacrylate phosphate (CAS # 52628-03-2, Figure 3). This UV-curable compound commonly is referred to as HEMA Phosphate.
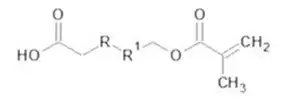
The methacrylate functionality allows the molecule to participate in the photopolymerization reaction binding the HEMA Phosphate structure into the resulting photopolymer. The phosphate functionality then can complex with metal ions on the surface of the substrate, resulting in the bonding of the coating or ink to the metal surface. An additional benefit is an improvement in corrosion resistance. The basic structure illustrated in Figure 3 is not limited to phosphates only. If the phosphate group is substituted by a carboxylic acid group, a class of adhesion promotors commonly referred to as acid functionalized adhesion promoters are obtained (Figure 4). The mechanism of adhesion promotion for these systems is like that of HEMA Phosphate.
Adhesion will continue to play a critical role in a wide range of UV and EB curing applications. Fortunately, as can be seen in this brief overview, there are many different approaches for improving adhesion. By evaluating one or more of these approaches in a project, most adhesion concerns can be “unstuck”!
 Guest Columnist:
Guest Columnist:
Mike Idacavage
Founder, Radical Curing, LLC
mike.idacavage@radicalcuring.com