By Curtis Lustig and Srinivasan Chakrapani, IGM Resins
The use of UV-curable coatings continues to expand into applications that traditionally have been the domain of other coating technologies. UV-curable materials offer the advantages of faster cure times, shorter production lines, greater energy savings and a reduced environmental impact. UV-curable materials now are found in a number of different applications. These include wood coatings, vinyl flooring, metal coatings, flexible packaging films and electronic assemblies, as well as numerous other applications. However, some of the applications involve challenging substrates and require more specialized formulations utilizing special functionalities capable of achieving optimal substrate adhesion and the desired final performance. The inclusion of UV-compatible adhesion promoters for challenging substrates could be an option in these cases. This article will look at some of the acrylate materials that may be helpful in obtaining optimum adhesion. In addition, some of the advantages accompanying the advent of UV LED curing technology will be discussed.
Substrates utilized
One of the most important flexible substrates used in the industry is biaxially oriented polypropylene (BOPP). It is mechanically rugged and has a high chemical resistance.1 It is used in a variety of roles, including packaging and adhesive tape applications. However, due to their low hydrophilicity, low surface energy and lack of functional groups on its surface, these films pose a challenge for inks and adhesives for durable coatings.
Polyethylene terephthalate glycol (PETG) is a transparent, high-gloss film used in medical, food packaging, thermoforming and electronics applications. It is a modified (by copolymerization) version of PET, which was patented in 1941 and later given the trademark Mylar by E.I. DuPont.2 By design and application, it is heat sensitive. Due to the smoothness of the PETG surface, it can be difficult to bond to.
Cast polypropylene film, usually known as CPP film, is used widely in packaging. It is one of the most common films used for metalization.3 CPP films are used in food and confectionary, oil and lube, detergent and shampoo, textile, pouch and bag, pharmaceutical tablet, disposable syringe, needles and medical appliances blister packing. Due to its low surface energy, adhesion can be a challenge.
Shown in Figures 1 through 5 are the FTIR spectra of both surfaces of the five substrates. They reveal that one side of each substrate, except for PETG, has been treated to improve adhesion. The treatment involved the application of a proprietary primer to the films.
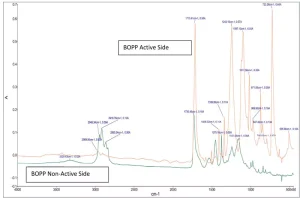
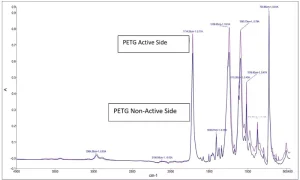
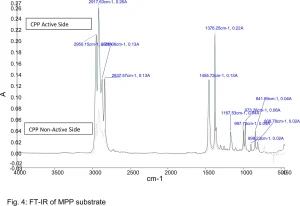
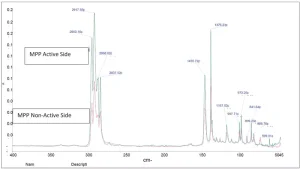
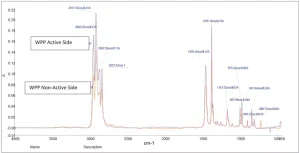
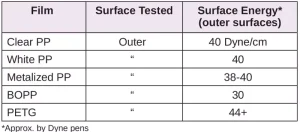
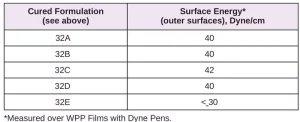
Surface energy is a key parameter in determining the wettability of a substrate. Lower surface energy materials will wet higher surface energy substrates. Table 1 provides the reported surface energies of some common polymer substrates. Table 2 shows the surface energies of the substrates, as they were measured for this evaluation.

UV-curable materials tested
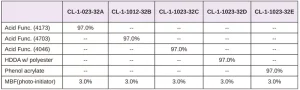
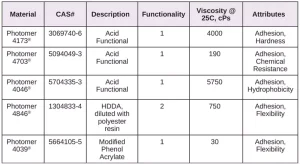
The adhesion of five different photomer acrylate materials (from IGM) to the polymer films of interest were evaluated in this study. One of their key attributes is adhesion improvement.
Coating process

As shown in Figure 6, the films were attached to steel Q-panels and then coated with #4 Draw Down Rod (for a film thickness of approximately 1 mil/25 microns) using the draw down technique. The films were kept free of dust but were not cleaned prior to being coated.
Curing process

A Fusion UV oven equipped with a Type H mercury vapor bulb (Figure 7) was used to cure the coatings. Samples were passed through the UV oven three times at a line speed of 63 fpm (approx. 145 mJ/cm2). The coatings were cured immediately after application.
Test methods
- Tape adhesion4: The standard tape test using 3M Brand 600 tape in which a length of tape (2.0″ minimum) was applied to the sample, pressed to remove entrapped air and then rapidly and forcefully removed (at a right angle to the test area). A new, unused strip of tape was used for each test.
-

Figure 8. Visual of tape on crease test: left, passed – PH4046® on MPP; right, failed – tape on crease – PH4173® Tape adhesion on folded sample: The coated film sample was folded over 180 degrees. Any cracking or delamination at this point was noted. Then 3M Brand 600 tape was applied to the folded area and forcefully removed. Any lifted or peeled coating was noted (Figure 8).
- Crosshatch tape test5 (not used on film samples): A standard multiblade cutter was used to make two perpendicular sets of cuts into the coating. Any flaking or peeling was noted. Then a strip of 3M Brand 600 tape was applied. The entrapped air was pressed out and then the tape was removed forcefully. Any lifting or peeling of the coating was noted.
Results
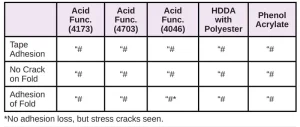
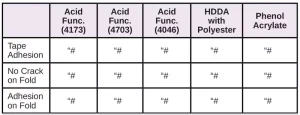
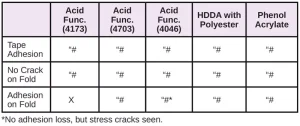
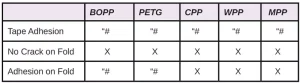
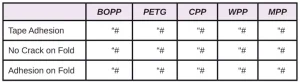
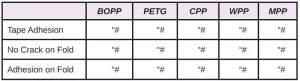
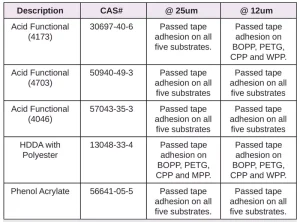
In Tables 6 through 10, a hash mark ““#” indicates that the sample passed the evaluation. An “X” indicates failure.
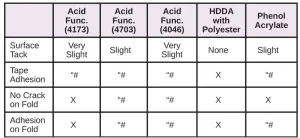
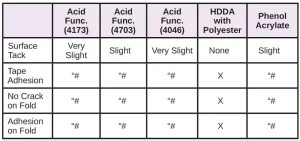
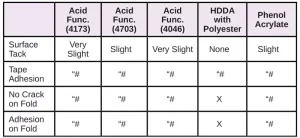
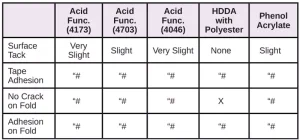
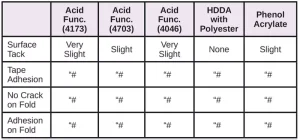
In the next evaluation, the coatings were applied at a thickness of about 12 microns. Though there were some differences, particularly for the WPP and MPP films, the results were similar overall. See Tables 11 through 15.
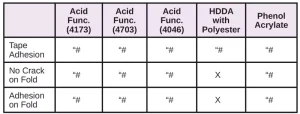
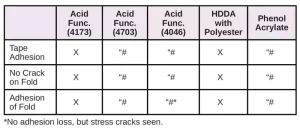
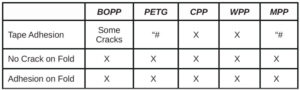
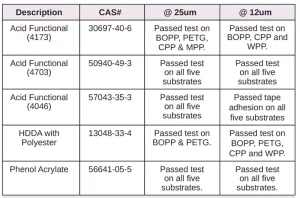
For comparison, several other materials – Photomer 4226 (Dipropyleneglycol diacrylate), Photomer 4017 (Hexanediol diacrylate), Photomer 4034 (HECLA) and Photomer 4184 (2[[Butylamino) carbonyl] oxy] ethyl acrylate) – were evaluated on the five films. As shown in Tables 16 through 19, PH4034 and PH4184 performed very well. Though HDDA performed well in the tape test on the films, overall, PH4226 and PH4017 did not perform as well as the five materials specifically recommended as adhesion promoters.
Coatings on glass

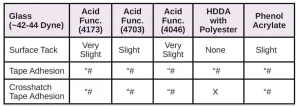
Glass sometimes can be a challenging substrate, particularly if it is treated with tin-oxide. Unfortunately, due to the unavailability of suitable samples of this material, a common craft-type glass was used for this evaluation. Tape adhesion5 and crosshatch tape adhesion6 tests were performed, with the results shown in Table 20 and Figure 9.
Summary
As noted, all the adhesion-promoting materials performed better, overall, than the control materials (DPGDA and HDDA) on the packaging films.
The results for each adhesion promoting acrylate are summarized in Tables 21 and 22. These materials may be used as adhesion-promoting additives in UV formulations for packaging materials or they may be used by themselves, where suitable, as a primer layer.
References
- Adapted from Adhesion and Adhesives: Science and Technology; Anthony J. Kinloch, New York: Chapman and Hall (1987).
- Kenneth S. Whiteley, T. Geoffrey Heggs, Hartmut Koch, Ralph L. Mawer, Wolfgang Immel (2005). “Polyolefins.” Ullmans’s Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH.
- Whinfield, John Rex and Dickson, James Tennant (1941) “Improvements Relating to the Manufacture of Highly Polymeric Substances,” UK Patent 578,079.
- Mount, E. (2004) Metalized Films for Food Packaging. Converting Magazine vol 3.
- ASTM Tape Adhesion Strength Testing, ASTM D3330.
- ASTM Standard Test Methods for Measuring Adhesion by Tape Test, ASTM D3359.






