By Carla Brown, manager, corporate communications – circular economy specialist, Siegwerk Druckfarben AG & Co.
A note from RadTech:
The RadTech Sustainability Committee is embarking on our group’s sustainability journey, and we thank Carla Brown, manager, Corporate Communications – Circular Economy Specialist, Siegwerk Druckfarben AG & Co., for sharing her work in a series of modules UV+EB Technology will publish throughout the year. This is the fourth and final installment.
Migration occurs when substances move from a printed layer into the packed food due to their chemical characteristics and molecular size. In this lesson, we will have a closer look at some aspects related to migration.
Food packaging examples, risks and problems

As explained in previous lessons, using a standard UV ink system for food packaging will very likely cause unwanted migration into the packed food. That’s why specific inks are required when food packaging is printed – these are called low migration (LM) or migration optimized inks. The risks associated with certain packaging types are explained below (Images 1-3).
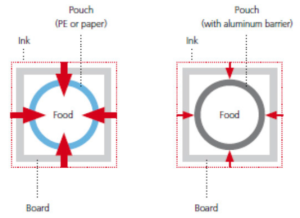
Pouches for dry food
Risk and problems without LM products:
- Possible set-off risk due to reel-to-reel production
- Low diffusion migration risk because of aluminum with high-barrier function
- Degradation products are able to migrate into the PE layer of the pouch
In-Mold Labels and Self-adhesive Labels on Cups
Risk and problems without LM products:
- Possible set-off due to storage in stack
- Low diffusion migration risk because of cup material thickness
Shrink Sleeves PP, PE or PS and Shrink Sleeves PET
Risk and problems without LM products:
- Diffusion migration possible > low – medium
- Depending on storage time and thickness of bottles, there is a risk of direct migration
How does migration occur?
Three different types of migration exist: set-off migration, diffusion migration and gas phase migration.
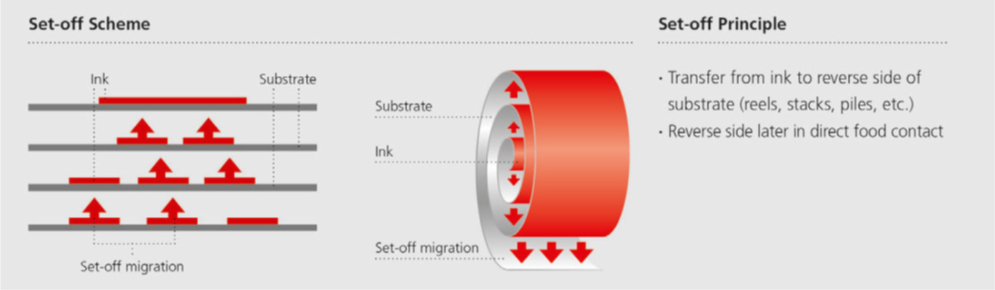
Set-off migration
Migrants can migrate from one layer to another, such as from a surface-printed layer to the non-printed food-contact surface, which is brought into contact with food later on. If these are in direct or close contact, like in a reel or a stack after printing, set-off migration can occur due to the pressure existing in the reel or stack (Image 4).
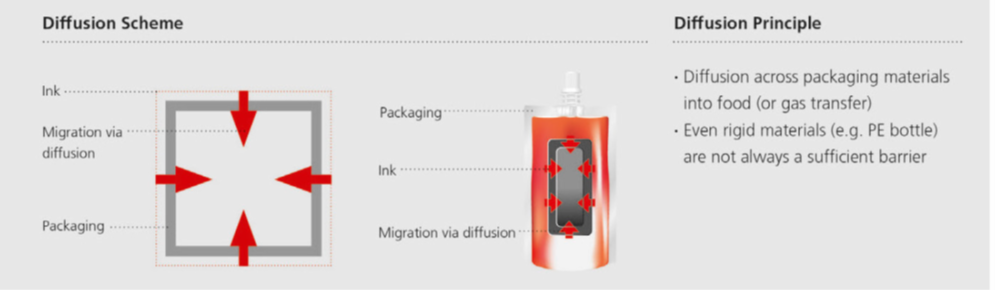
Diffusion migration
Small and mobile molecules easily can penetrate into and diffuse across packaging material layers. This can occur if the printed material has not yet been converted into a food package and filled with food, or later on when the printed package is filled with food and the food starts to extract the migrants from the packaging material (Image 5).
Gas phase migration
Migrants also can migrate from the substrate via the gas phase within the pack to end up in food, which acts as a ‘recipient reservoir.’ These migrants can be mineral oils or some UV photoinitiators that might not be generally known as being volatile, such as organic solvents.
What is unique about a low-migration system?
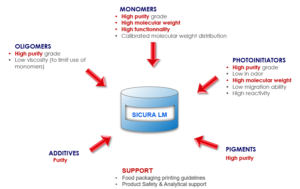
There is a clear difference in the concept to formulate standard non-LM inks and LM inks. It is the molecular weight of the components used. The rule of thumb says that a component with a molecular weight above 1000 Dalton (= atomic mass unit, g/mol) will not migrate. Hence, for food packaging inks, high molecular weight photoinitiators, monomers and oligomers that possess a low tendency to migrate, typically are used (Image 7).
Regulations overview
The following represents an overview of regulations in different regions (Chart 1).
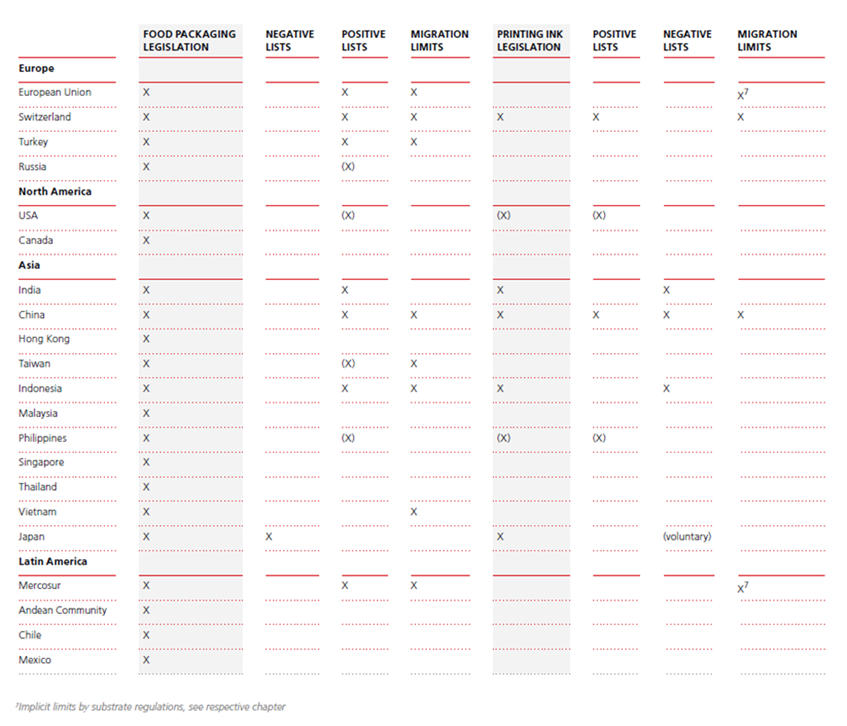
Europe, Middle East, Africa (EMEA)
Regulation (EC) No 1935/2004 (“Framework Regulation”)
- Important: Article 3: No endanger of human health, no unacceptable change in the composition of the food, no deterioration in the organoleptic characteristics thereof
Regulation (EC) No 2023/2006 (“GMP Regulation”)
- Important: Effective quality management system, printed surface shall not come into direct contact with food, migration and invisible set-off below limits
Regulation (EU) No 10/2011 (“Plastic Regulation”)
- Important: Only for materials made of plastic, positive list of evaluated substances, rules for migration testing, Overall Migration Limit (OML) = 60 mg/kg food, Specific Migration Limit (SML) for single substances in mg/kg food, Risk Assessment for not listed substances to prove compliance with Art. 3 of Framework Regulation, declaration of compliance for the final product mandatory
Swiss Ordinance 817.023.21 (“Ordinance of the FDHA on Materials and Articles intended to come into contact with Food”)
- Important: First positive list for printing inks worldwide, in scope are only intentionally added substances (IAS) and inks for non-DFC applications, toxicological evaluated substances in List A with SML or OML (i.a.), non-evaluated substances in List B with a detection limit of 10 ppb
German Ink Ordinance (“21st Ordinance amending the consumer goods ordinance”)
Important: Positive list for printing inks, in scope are IAS and inks for non-DFC as well as DFC applications, unlisted substances are allowed but not for DFC applications and not classified as “CMR” and only with a migration below 10 ppb, NIAS can be self-evaluated in accordance with internationally recognized scientific principles on Risk Assessment, transition time of four years (i.e. applicable from January 1, 2026)
Canada, United States (CUSA)
United States: FDA section 21 CFR 110.80 (“Current Good manufacturing practice in manufacturing, packing, or holding human food; Processes and controls“)
- Important: Corresponds to a “Framework Regulation”
Canada: Food and Drugs Act (R.S.C., 1985, c. F-27)
India
Indian Standard IS 15495:2020 (“Printing Ink for food packaging – Code of practice”)
- Important: Exclusion list for substances from printing ink formulations in Annex A
China
National Standard GB 4806.1-2016 (“General Safety Requirements of Food Contact Materials and Articles”)
- Important: Corresponds to a “Framework Regulation”
National Standard GB 9685-2016 (“Standard for Uses of Additives in Food Contact Materials and Articles”)
- Important: Additives for printing inks listed in chapter A4
Southeast Asia (SEA)
Regulation 20/2019 on Food Packaging (Indonesia)
Law No. 55/2010/QH12 on Food Safety (Vietnam)
Republic Act No. 10611/2013 (Philippines)
Latin America (LATAM)
GMC MERCOSUR RES. N° 03/92
- Important: Corresponds to a “Framework Regulation”
GMC MERCOSUR RES. N° 56/92 (“General Provisions for Food Contact Plastics”), last amended with No 20/21
GMC MERCOSUR RES. N° 02/12 (“Plastic Regulation”), last amended with No 19/21
Testing regulations
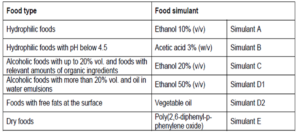
According to Annex III of the Regulation (EU) No 10/2011, the best suited of the food simulants shall be used for migration testing. These are noted in Chart 2.
Migration testing
As migration testing requires a long and complex analytical process, it is not routinely performed as a quality control test. To avoid complexity and manage timing, a sampling strategy and a pre-validation process for checking migration compliance are helpful. Given the wide variety of packaging types and structures, and given the variance in printing and converting conditions, a sample selection strategy is advised. The goal is to select the different families of samples of packaging that are covering the whole printer’s food packaging production. Applying a worst-case scenario approach always is recommended to minimize the risk of non-compliance and to maximize the window of compliance.
Worst-case scenario applies for many parameters, such as substrate thickness, ink coverage, surface volume ratio, printing conditions (e.g. press speeds, configuration of UV curing), storage and converting conditions.
The representativity of the sample for migration testing is essential. For example, storage conditions (pressure, temperature and time) of the samples need to be taken into consideration. It obviously is preferrable to test packaging that has been stored in industrial conditions, especially when potential migration by set-off needs to be evaluated.
During sampling handling, precautions have to be taken to avoid any changes or damage of the samples, which would affect the properties. It is, for example, recommended to wrap the sample in plain aluminum foil to prevent any relevant interaction with its environment during transport. A complete traceability from the sample (identification and labeling) to the test result also should be ensured (including inks used and printing parameters).
How testing is conducted

In a first step, the printed sample is exposed to simulant either using a standard cell or directly filled in the packaging (Images 8 and 9). After the incubation time, the simulant is injected in a GCMS or LCMS (or both). These instruments can detect the migrants down to concentrations of ppb and below.
The resulting graphs are analyzed and the migration per component listed. Usually, detected quantities in µg/dm2 need to be multiplied by six to be converted to µg/kg (= ppb), according to the EU-convention of 6dm2 packaging for 1kg food.
Further information in regard to migration testing are given in the “EuPIA Guidance on Migration Test Methods for the evaluation of substances in printing inks and varnishes for food contact materials,” which can be downloaded on the EuPIA webpage at
www.eupia.org.






